Center for Radiology Research Resources (CR3)
The Center for Radiology Research Resources (CR3) was created to provide a clear and efficient path for researchers to partner with the Department of Radiology when a study requires imaging services. CR3 provides study teams across the University and M Health Fairview with recommendations for enhancements to the proposed protocol and insights on technological requirements.
CR3 Scope of Services
CR3 will assist clinical trials, with human subjects, that have imaging scans planned at any of the M Health Fairview imaging locations. Additionally, CR3 will provide protocol review or consultation services to any clinical trial with regard to their imaging needs, regardless of the location of the planned scans.
CR3 Rates
See full rate list at bottom of page.
CR3 Services
CR3 will provide a wide array of services to help support your research, including:
- Reviews of research protocols and imaging manual to ensure we can provide the services needed at the locations requested
- Recommendations to enhance protocols
- Protocol-specific research read reports
- Consultations with radiologists
- Technology recommendations
- Location options for imaging services
- Assistance with forms related to imaging technology
- Assistance with identifying correct CPT codes for imaging studies
- Assistance with image de-identification and transfer
Contact for General Questions
radresearchread@umn.edu
CR3 Intake Form
To begin onboarding for new study
Image De-Identification and Transfer Request Form
For use only after your onboarding is complete
Faculty and Staff
CR3 Leadership

Charles A. Dietz, Jr., MD
Radiology Department Chair
CR3 Director
Contact
dietz004@umn.edu

Patrick Bolan, PhD
CR3 Associate Director
Contact
bola0035@umn.edu
Protocol Review Committee
Zuzan Cayci, MD
Kelly Dietz, MD
Noelle Hoven, MD
Can Ozutemiz, MD
Takashi Takahashi, MD
Rajesh Thampy, MD
CR3 Staff

Megan Leland
Imaging Manager, CR3 and CCIR
Contact
mleland10@umphysicians.umn.edu

Christine Damases Kasi
Radiology Research Technologist
Contact
cdamases@umn.edu

Jenavieve (Jenna) Olson
CR3 Coordinator
Contact
olso7106@umn.edu
612-626-5546
How to Request CR3 Services
Request Services for New Studies
You can begin working with CR3 as soon as you begin planning your study, if radiology support is needed. Complete the CR3 Intake Form to begin the process. A CR3 coordinator will reach out to you to begin a service agreement and collect the protocol documents and imaging manuals for review.
Image De-Identification and Transfer
Once you have completed your CR3 Intake/Onboarding process and have requested Imaging De-Identification and Transfer Services, use the Image Transfer Request Form to submit requests. Once we receive a request, we will typically have your images anonymized and transferred to your Box folder or uploaded to the sponsors (depending on the service you selected) within 5 business days. If you have a specific deadline, please indicate that on the form.
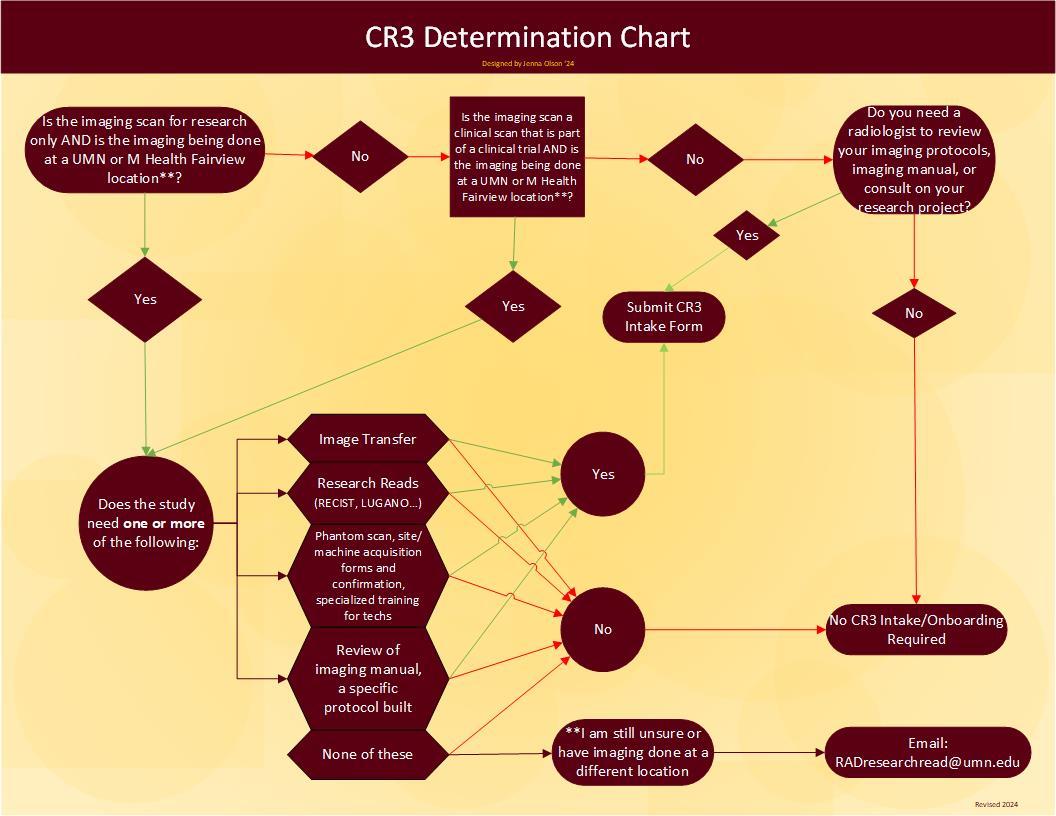
Do I need CR3 Services?
Unsure if you need CR3 Services? Use this decision tree to help determine if you need to submit an intake form or not. If your unique research issue isn't addressed in this model, just contact radresearchread@umn.edu.
CR3 Rates
Internal Rates
Internal rates are for funding routed through the University with a sponsored or non-sponsored chart string.
Rates last updated on 9/1/23.
Onboarding with Protocol Review: $750
The CR3 team will evaluate your study to determine if a protocol review is required; not all studies will need this service. If your study does require a protocol review, this one time fee will be billed through UMN and includes:
- Reviewing the protocol to assure we can provide the service requested
- Provide feedback and recommendations for enhancing the protocol
- Providing any start up documents/forms needed for study start-up
- Setting up protocols/study details in CR3 systems
Onboarding without Protocol Review: $145
The CR3 team will evaluate your study to determine if a protocol review is required; not all studies will need this service. If your study does not require a protocol review, this one time fee will be billed through UMN and includes:
- Providing any start up documents/forms needed for study start-up
- Setting up study details in CR3 systems
Research Read: $350 per criteria
This fee will be billed in two separate portions. $150 will be billed to the study through FV Research Administration. $200 will be billed through UMN. This fee includes:
- Research read/report to meet specific research protocol needs (this is an additional read, beyond the standard of care read)
- CR3 staff support to aid with special requests, troubleshoot issues, answer questions, meet timelines
- CR3 staff will facilitate/schedule meetings with radiologist in regard to questions about report findings
Image De-Identification and Transfer to Box Folder: $40 per imaging scan
This fee includes:
- De-identification and uploading patient scans to a secure Box folder shared to the study team
Imaging De-Identification and Transfer to Study Sponsor: $65 per imaging scan
This fee includes:
- De-identification and uploading patient scans to a study central site
Please note: Fee subject to change with a minimum of 30 days notice to existing clients
External Rates
External rates are for standard billable services to external customers. A contract must be in place prior to use.
We are currently only offering external contracts for CR3 services to Fairview Health Services.