Collaborative work aims to correct cellular dysfunction in the injured brain
Professor Walter Low, PhD, paints a nurturing picture of certain cells in the brain that help you recover after a traumatic brain injury (TBI). The cells, called microglia, play an important role in maintaining connections between neurons in the brain – but also can be destructive if over-activated following TBI.
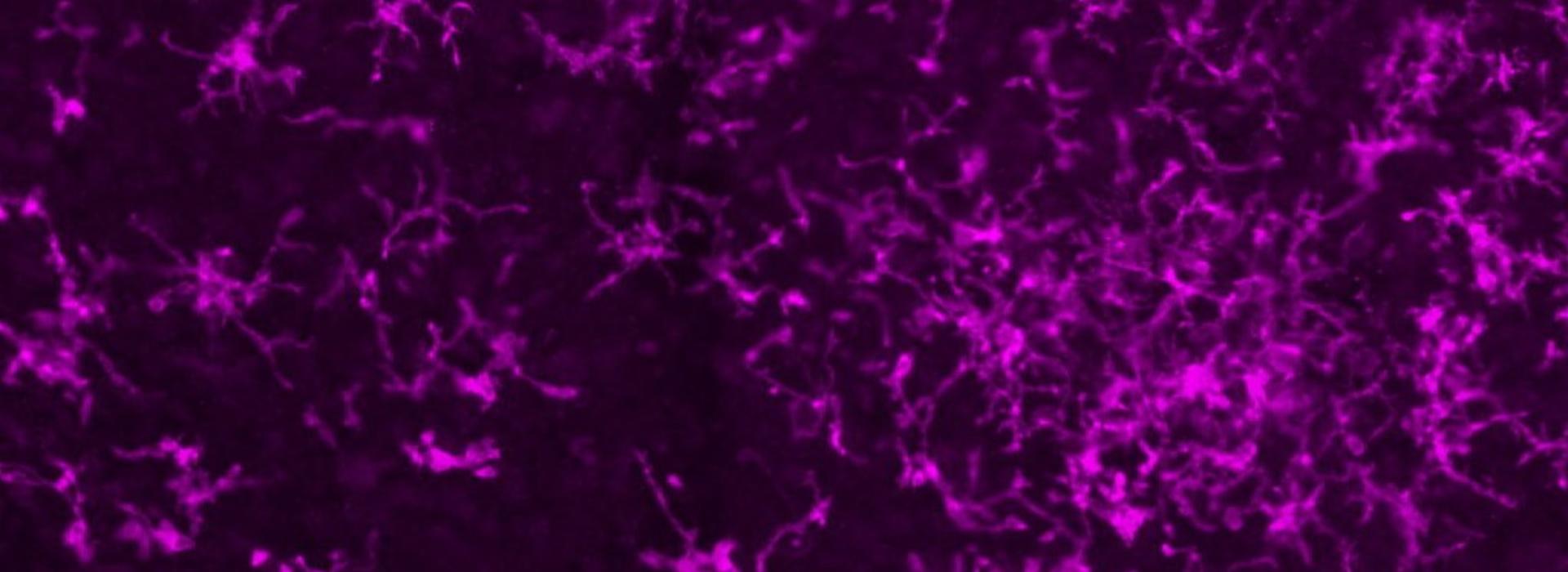
“Microglia cells play several roles in the brain,” said Low. “When you look at them under the microscope [see above], they extend their filopodia and touch synaptic connections. In what amounts to a nurturing response, they maintain that connectivity, which helps support the neural circuitry in the brain.”
Brain's immune system
When there is an infection, Low noted that these cells also act like the brain’s immune system. “If they detect that something is wrong, they switch their role to try and destroy the invaders,” he said. “Their nurturing role becomes an inflammatory one.” As the cells continue to fight the infection, they can rip apart the synaptic connections they once nurtured.
This change in role is important to consider in traumatic brain injury – and the subject of a new three-year study being led by Low in collaboration with U of M colleagues, Andrew Grande, MD, and Ann Parr, MD, PhD, Neurosurgery Department, Maxim Cheeran, MVSc, PhD, Department of Veterinary Medicine (focus: immunology); Wei Chen, PhD, and Xiao-Hong Zhu, PhD, Department of Radiology (focus: brain imaging), and colleagues from the Uniformed Services University (USU) of the Health Sciences, Bethesda, MD.
U of M influence
Thanks in part to the influence and hard work of Greg Beilman, MD, of the U’s Department of Surgery, Congress recently included $35M of funding for a new partnership between Minnesota (University of Minnesota Medical School, Medical Alley, and Fairview) and USU. According to Beilman, the resulting grant program is aimed at health issues that impact the military – which are also important to Minnesota communities – and where the strengths of the state’s alliance can be best applied.

The UMN/USU study led by Low (pictured here) is titled, "Microglial Replacement for Treatment of Traumatic Brain Injury," and is one of 13 from a total of 46 full grant applications that were funded following a peer-reviewed process. Applications associated with traumatic brain injury were given priority as recent research indicates that slightly more than 20 percent of Veterans serving after September 11, 2001, have been exposed to either mild (17.5 percent) or moderate to severe TBI (3 percent)1.
Replacing dysfunctional cells
Low and his team aim to deplete dysfunctional microglia in brain-injured rats and mice and replace them with exogenic microglia or stem cells that will become functioning microglia. “Many TBI patients go on to have prolonged neurological problems because of persistent brain inflammation,” said Low. “Key players in this inflammatory environment are microglia cells. As they get older, the cells become compromised and begin to engulf the synaptic connections between neural cells. This creates a disconnect and results in cognitive problems.”
The team will take induced pluripotent stem cells (iPSC), which can be differentiated into any kind of cell in the body and change them into hematopoietic stem cells — precursors to microglia. “The general application will require off-the-shelf cells that are a close match to the recipient,” said Low. “These microglia are young and won’t have the phenotype of overactive cells seen in an injured brain.”
Stem cell experience
Both Grande and Parr have experience working with iPSC translation; Grande in the treatment of stroke and Parr in treating spinal cord injury. “Dr. Grande and Dr. Parr will help us translate the research to the clinical setting and are assisting with different aspects of the study,” said Low. “Being involved now will help them understand how these cells can be manipulated in the future.”
The team’s expectation is that one treatment will be all that’s needed. “The new microglia will take over the deficient cells,” said Low. “In previous studies, functional benefits arose one to two months after replacement in an animal model. We expect that it might take a little longer in humans.” The team will know they’ve successfully transformed the brain environments of their brain-injured animal subjects when they demonstrate the ability to walk again and to navigate certain mazes.
Low and his team hope to get started on this work as soon as the budget is finalized with the funding agency.