Participating in a clinical trial — a journey from the edge of darkness and inferno to light and promise
Julie Nitz feels incredibly fortunate to have connected with former Department Head Clark C. Chen, MD, PhD, and as a result, to be enrolled in one of the clinical trials he is leading at the U of M.

Julie (pictured here) was diagnosed last year with glioblastoma – the most common and deadliest of adult brain cancers. After Chen performed her tumor resection in August, she began her journey as a member of the GESTALT (GammaTile Enhanced Stupp ALTernative) clinical trial for patients with newly diagnosed glioblastoma.
Period of growth
One of the reasons that glioblastoma is so deadly is because the cancer is characterized by a high density of microscopic brain cancer cells immediately adjacent to the tumor that the surgeon can see. During surgery, the visible tumor is removed; however, the microscopic cancer cells continue to grow during the four to six weeks required for the patient to recover before chemotherapy and radiation can continue their treatment.
Note: the current standard of care for glioblastoma that includes chemotherapy and radiation therapy is often referred to as the Stupp protocol, named after the oncologist who developed it.
“Between 50 and 90 percent of patients suffer significant tumor growth during this recovery period after the initial surgery,” said Chen. Such growth diminishes the effectiveness of the subsequent therapy and may lead to additional tumor growth and repeat surgery.

“To understand why, imagine the cancer cells as villains and the chemotherapy and radiation therapy as the heroes trained to stop them,” explained Chen (pictured here). The surgeon’s job is to remove as many villainous cancer cells as possible without harming the patient, thereby enhancing the effectiveness of chemotherapy and radiation therapy, Chen noted.
“If the surgery removed 900 cancer cells but the remaining 100 successfully recruited 900 more during the four to six weeks of recovery, the surgery is rendered futile and the subsequent chemotherapy and radiation therapy are unlikely to be effective,” said Chen. “Many patients are referred to me for repeat surgery because the glioblastoma grew significantly during the recovery period, leading to multiple brain surgeries during a relatively short period of time. Such news is devastating for the patient.”
Novel strategy
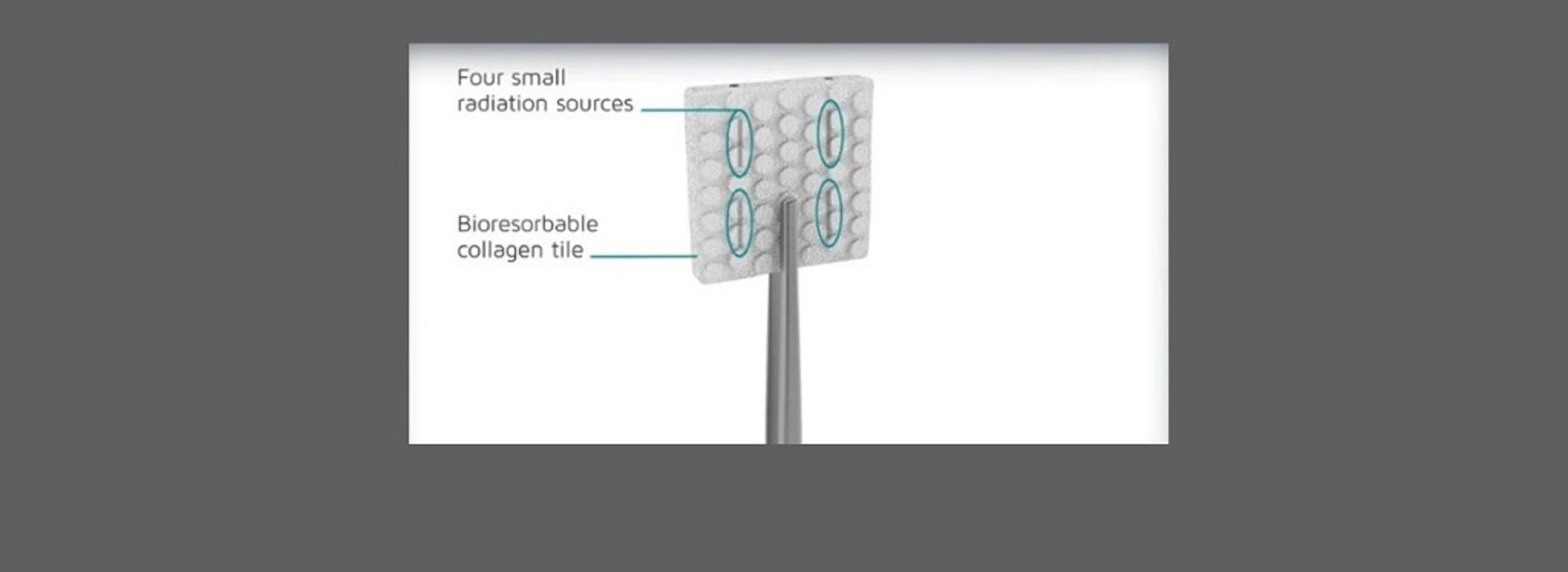
To prevent tumor growth during the recovery period, Chen developed a novel strategy involving surgical implant of what is known as GammaTile™. “GammaTile consists of resorbable collagen embedded with Cs-131 radiation sources and is implanted at the time of surgery,” Chen explained (see illustration above, courtesy of GT Medical Technologies). “When implanted, the patient immediately receives radiation therapy, avoiding the four to six weeks of recovery during which the tumor cells grow unimpeded.”
In collaboration with GT Medical, the manufacturer of GammaTile, Chen spearheaded its clinical development and use for glioblastoma patients. GammaTile for brain tumor patients was cleared by the FDA in 2019, and Chen was the first in the world to implant it in a glioblastoma patient.
Developed the study
To firmly establish the safety and effectiveness of GammaTile for glioblastoma patients, Chen worked with GT Medical to develop the GESTALT clinical study. U of M was the first institution in the United States to enroll for this study, and Julie was the first patient enrolled.
When Julie first learned of her diagnosis, she called her brother, Bob, who is also a physician. Understanding the aggressive nature of glioblastoma, he had painted a dire picture for Julie; however, meeting with Chen changed Bob and Julie’s perspective. “Dr. Chen gave me his cell phone number and we had several conversations to help me understand what was going to happen,” Julie said.
Recent MRI clear
Chen explained the benefit of GammaTile to Julie and Bob and that participating in the GESTALT trial would give her an opportunity to beat this deadly cancer. “The hope and optimism this kind of option gives an anxious patient like Julie cannot be underestimated,” said Chen. “The reality is supporting the trial’s theory. Julie had her eight-month MRI recently and it showed no evidence of tumor growth. In previous patients who were treated without GammaTile, more than 70 percent would have shown significant tumor growth by now.”
Julie feels like she’s doing well. “Dr. Chen called me to say that my last MRI looked really good,” she said. She wishes there was a better way for people to find out about studies like the one in which she is enrolled. “If I hadn’t talked with Dr. Chen, I wouldn’t have known about it,” Julie said. “I have an acquaintance who has the same thing I do. She wasn’t enrolled in a study and the last time she went to see her care team they told her she had a year to live. I’m so glad I went to Dr. Chen.”
Best of times, worst of times
Julie takes a “best of times, worst of times” approach to her diagnosis and the care she is receiving at the U. “Other than it being a horrible experience, it has been a good experience,” she said.
“Pastor Charles Spurgeon famously observed, ‘Faith goes up the stairs that love has built and looks out the windows which hope has opened,’” noted Chen. “It is hope that allowed Julie and Bob to catapult from darkness and inferno to light and promise. We must continue to further our science, as it is the spring of hope eternal.”