Study evaluates time-tested neurostimulation technique for use in treatment-resistant depression
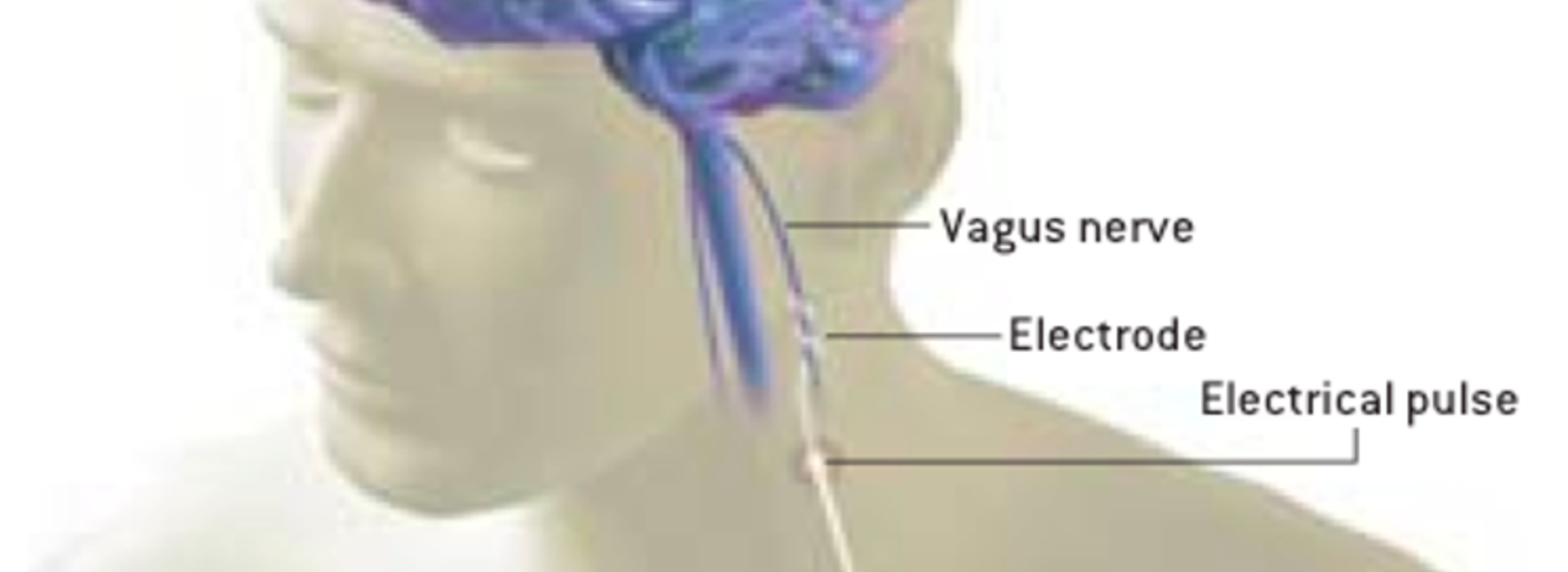
Clinicians have been stimulating the vagus nerve to treat epilepsy symptoms since the procedure received FDA approval in 1997. In 2005, the agency approved the use of an implanted vagus nerve stimulator for treatment-resistant depression (TRD). Even though approved for this use, the Centers for Medicare and Medicaid (CMS) has deemed it experimental and would not pay for the procedure. Insurers have followed CMS’ lead.
Re-vitalized industry interest in demonstrating the “non-experimental” nature of vagus nerve stimulation (VNS) for treating TRD has resulted in an unusual CMS/industry-funded multicenter study known as RECOVER. The University of Minnesota is one of almost 60 sites throughout the country helping implant vagus nerve stimulators in 1,100 study participants to ascertain its long-term clinical efficacy in treating refractory depression.
Psychiatrist Ziad Nahas, MD, MSCR, of the Department of Psychiatry and Behavioral Sciences, is the principal investigator (PI) for the U’s portion of the study. Neurosurgeons Michael C. Park, MD, PhD; Robert McGovern, MD; and David Darrow, MD, MPH, of the Neurosurgery Department, are implanting the vagus nerve stimulators. Learn more.