Treating & Preventing COVID-19
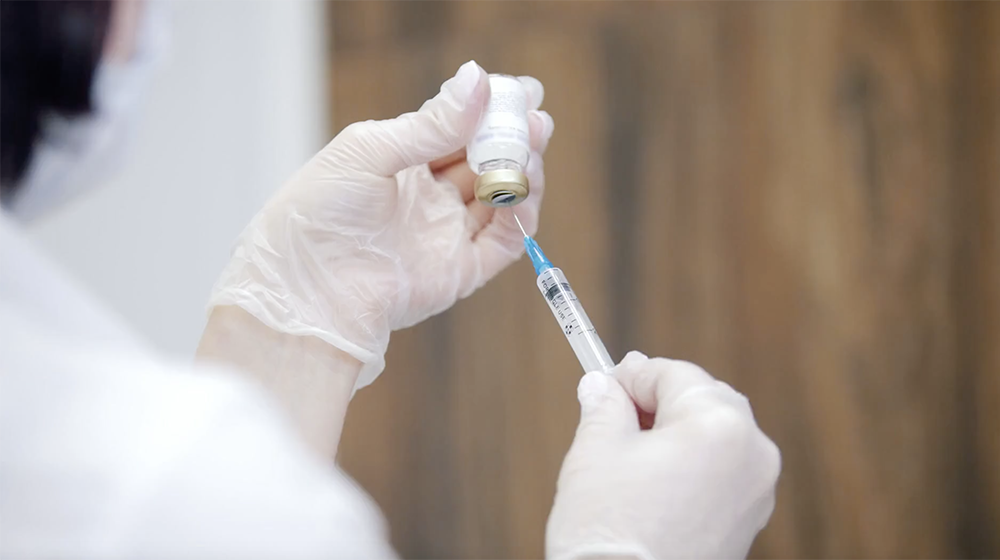
University of Minnesota Medical School launches COVID-19 vaccine trial
The University of Minnesota Medical School announced today its plans to participate in a Phase 3 COVID-19 clinical trial in the fight against the novel coronavirus. The clinical trial will test the safety and effectiveness of an investigational vaccine developed by U.S. biotechnology company Novavax, Inc., called NVX-CoV2373. Novavax is a late-stage biotechnology company developing next-generation vaccines for serious infectious diseases.
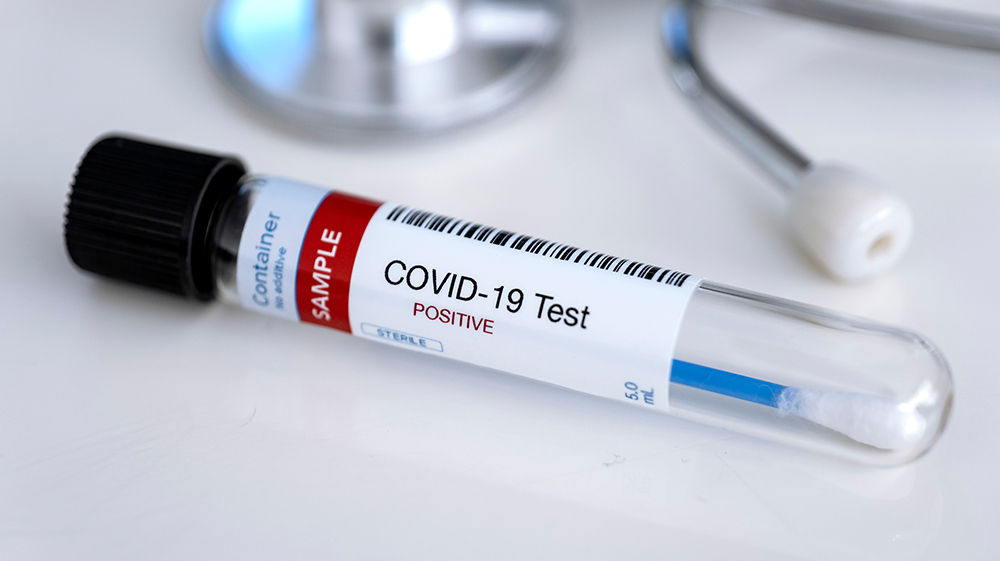
University of Minnesota Medical School researchers develop two new rapid COVID-19 diagnostic tests
University of Minnesota Medical School researchers have developed two new rapid diagnostic tests for COVID-19 – one to detect COVID-19 variants and one to help differentiate with other illnesses that have COVID-19-like symptoms. The findings were recently published in the journal Bioengineering.
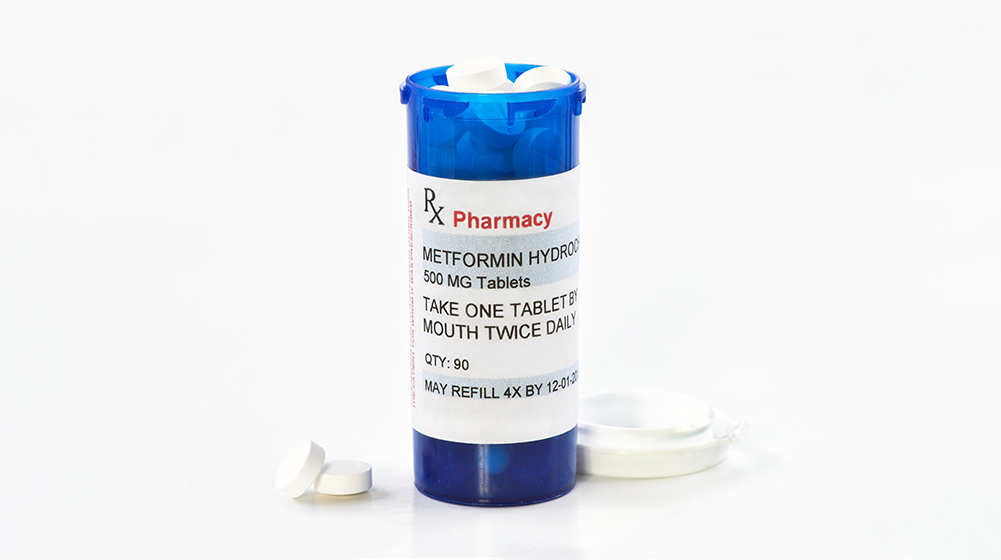
University of Minnesota Medical School launches clinical trial studying metformin treatment for COVID-19
A new multi-site clinical trial led by the University of Minnesota Medical School is studying the effectiveness of metformin, a generic medication for type 2 diabetes, in the treatment of COVID-19. It will be the first randomized clinical trial for COVID-19 in the world to include pregnant women.
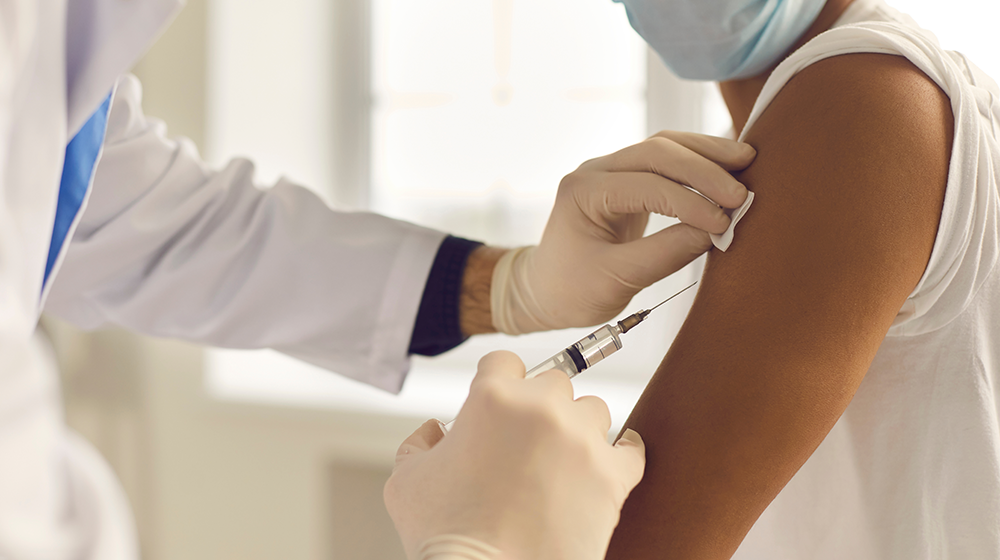
U of M Medical School launches trial to study COVID-19 vaccine efficacy among 18- to 29-year-olds
The University of Minnesota Medical School has joined a national study working to understand how well COVID-19 vaccines prevent infection and the spread of SARS-CoV-2 among young adults between the ages of 18 and 29. It is the only trial site in the four-state border region — among 40 sites in the nation — and is currently enrolling interested study participants in the Twin Cities.